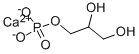
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt
Glycerophosphoric acid calcium salt
CAS: 27214-00-2;28917-82-0
Molecular Formula: C3H7CaO6P
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Names and Identifiers
| Name | Glycerophosphoric acid calcium salt |
| Synonyms | neurosin dicalcium Glycerophosphates Calcium glycerophosphate Calucium Glycerophosphate Calcium Glycerophosphoric Calcium glycerinophosphate CALCIUMGLYCEROPHOSPHATE,FCC 2,3-dihydroxypropyl phosphate CALCIUM GLYCEROPHOSPHATE POWDER Glycerol phosphate calcium salt (2R)-2,3-dihydroxypropyl phosphate (2S)-2,3-dihydroxypropyl phosphate Glycerophosphoric acid calcium salt calcium 2,3-dihydroxypropyl phosphate calcium 1,3-dihydroxypropan-2-yl phosphate [2-hydroxy-1-(hydroxymethyl)ethyl] phosphate calcium (2-hydroxy-1-methylol-ethyl) phosphate 2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt |
| CAS | 27214-00-2 28917-82-0 |
| EINECS | 248-328-5 |
| InChI | InChI=1/2C3H9O6P.2Ca/c4-1-3(5)2-9-10(6,7)8;4-1-3(2-5)9-10(6,7)8;;/h2*3-5H,1-2H2,(H2,6,7,8);;/q;;2*+2/p-4 |
| InChIKey | IWIRHXNCFWGFJE-UHFFFAOYSA-L |
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Physico-chemical Properties
| Molecular Formula | C3H7CaO6P |
| Molar Mass | 210.14 |
| Density | 1.82-1.855[at 20℃] |
| Melting Point | > 170℃ |
| Water Solubility | faint turbidity |
| Solubility | Sparingly soluble in water, practically insoluble in ethanol (96 per cent). |
| Vapor Presure | 0Pa at 25℃ |
| Appearance | powder to crystal |
| Color | White to Almost white |
| Merck | 14,1670 |
| Storage Condition | Inert atmosphere,Room Temperature |
| MDL | MFCD00042643 |
| Physical and Chemical Properties | Calcium glycerophosphate is a white crystalline powder. No odor, almost no odor, slightly bitter taste, moisture absorption. 130 deg C when the loss of crystal water to form nothing, more than 170 deg C decomposition. Dissolve in about 50ml of water per gram at 25 °c. When the temperature is lower, it is more soluble in water, and the aqueous solution is alkaline. Citric acid or lactic acid can improve its solubility in water. Almost insoluble in boiling water. Insoluble in ethanol. |
| Use | Nutraceuticals, for the preparation of compound preparations |
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Standard
Authoritative Data Verified Data
This product is by glycerol and phosphoric acid co-heat, and then neutralized with milk of lime, with ethanol precipitation, collect precipitation, after washing, drying and prepared. A mixture of 3-,D-and L-a-calcium glycerophosphate. Based on the dry product, the calcium content (Ca) should be 1 8.6% ~ 1 9.4%.
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Trait
Authoritative Data Verified Data
- This product is white to yellowish powder, odorless or odorless, with slight hygroscopicity.
- This product is slightly soluble in water and almost insoluble in ethanol.
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Differential diagnosis
Authoritative Data Verified Data
- take about O .lg of this product, dissolve it with 10ml of water and 10ml of dilute nitric acid, add 5 m l of ammonium molybdate solution, and boil, yellow precipitate will occur.
- take about O . lg of this product, add potassium hydrogen sulfate O. lg, mix it, put it in a test tube to heat it, and the irritating odor of acrolein will occur.
- This product shows the flame reaction of calcium salt (General rule 0301).
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Exam
Authoritative Data Verified Data
pH
take this product l .O g, dissolved with water 100ml, add phenolphthalein indicator solution 2 drops, with sodium hydroxide titration solution (0. ltn o l/L) or hydrochloric acid titration solution (0. lm o l/L) titration, consumption of sodium hydroxide titration solution (0. lm o l/L) or hydrochloric acid titration solution (0. lm o l/L) 1. 7ml.
clarity and color of solution
take this product l. The solution should be clear and colorless. In case of turbidity, it should be compared with No. 3 turbidity standard solution (General rule 0901, first method), not more concentrated.
chloride
take 0801g of this product and check it according to law (general rule). Compared with the control solution made of 5. 0 m l of standard sodium chloride solution, it should not be more concentrated (0. 02%).
sulfate
take 0802g of this product and check it according to law (general rule). Compared with the control solution made of 5. 0ml of standard potassium sulfate solution, it should not be more concentrated ().
phosphate
take this product l.O g, put in 25ml Nessler's colorimetric tube, add 10ml of dilute nitric acid to dissolve, add 10ml of ammonium molybdate test solution, shake well, and let stand for 10 minutes, such as color development, with phosphate standard solution (precision weigh potassium dihydrogen phosphate 0.192g, put it in a 100ml measuring flask, add water to dissolve and dilute to the scale, shake, take 3ml precision, put it in another 100ml measuring flask, add dilute nitric acid to the scale, Shake) 10ml of the control solution should not be more concentrated (0.04%).
limonate
take 5 g of this product and put it in a beaker, add 20M l of fresh boiling water, dissolve it, filter it, add sulfuric acid to the filtrate. 15ml, shaking, filtering, adding 5 m l of mercury sulfate solution to the filtrate, heating to boiling, adding 0.5tnl potassium permanganate solution, heating to boiling again, no precipitation should be generated.
free glycerol and soluble in fermentation broth
Take lg of this product, add 25ml of anhydrous ethanol, shake for 2 minutes, filter, filter residue is washed with anhydrous ethanol 5M l, combine filtrate and lotion, in an evaporating dish dried to constant weight at 70 ° C., it is evaporated on a water bath and dried at 70 ° C. For 1 hour. 5%).
loss on drying
take this product, in 1 5 0 1 drying 4 hours, loss of weight shall not exceed 12.0% (General rule 0831).
Iron Salt
take this product l.O g, with dilute hydrochloric acid 2M l and water 23ml after dissolving, according to the law (General Rule 0 8 0 7 ) , and standard iron solution 2. Compared with the control solution made of Oml, it should not be deeper (0. 002%).
Heavy metals
take this product l .O g, add acetate buffer (PH 3 .5) the amount of 2m l and water shall be adjusted to 2 5 m l, which shall be checked according to law (General Principles 0821, first law), and the heavy metal content shall not exceed 20 parts per million.
arsenic salt
take 0822g of this product, add 23ml of water and 5 m l of hydrochloric acid, dissolve it, and check it according to law (General rule 0.0003%, Law 1), which shall meet the requirements ().
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Content determination
Authoritative Data Verified Data
take this product 0. 2G, precision weighing, water 300tnl shaking to dissolve, add 10mo l/L sodium hydroxide solution 6. 0M l and calcium carboxylic acid indicator 1 5M g, with ethylenediamine tetraacetic acid disodium titration solution (0. 05mol/L) titration to the solution from purple to blue. Each lm of ethylenediamine tetraacetic acid disodium titration solution (0.05 mol/L) corresponds to 2.004 mg of Ca.
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Category
Authoritative Data Verified Data
pharmaceutical excipients, diluents and Hygroscopic agents.
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Storage
Authoritative Data Verified Data
sealed and stored in a dry place.
2,3-Dihydroxypropyl (dihydrogen phosphate), calcium salt - Reference Information
| LogP | -2.31 at 20℃ |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| application | calcium glycerophosphate is widely used in medicine, food, toothpaste and other industries. It can be used in food to play the role of calcium supplement; in medicine, it plays the biological and physiological role of calcium, has the function of perfecting tissue cells, forming bone tissue, and at the same time plays the role of calcium glycerophosphate metabolism, and has the function of supplementing brain vitality. |
| toothpaste additive | as early as 1960, calcium glycerophosphate was found to have antibacterial and anti-caries effects. Through research, it is found that it can increase the content of calcium and phosphorus in enamel, inhibit the demineralization of enamel, and improve the remineralization of enamel. Calcium glycerophosphate can change the acidity and alkalinity of enamel surface and enhance its acid resistance. The advantages of calcium glycerophosphate are self-evident, but there are not many applications in my country's oral care industry. Calcium glycerophosphate is an excellent oral product additive, especially suitable for calcium supplement toothpaste and chewing gum. It is also an ideal raw material for anti-caries additives. It can be used in children's toothpaste to promote tooth growth and calcium strengthening. The role of calcium glycerophosphate in toothpaste is mainly manifested in the following aspects: ①The control of plaque. ② Protective effect on dentin. (3) Influence on the ecological balance of oral flora. In addition, calcium glycerophosphate can improve the absorption of fluoride. 0.1% ~ 0.13% of calcium glycerophosphate is added to the toothpaste containing SMFP, which greatly improves the absorption rate of enamel to fluoride and strengthens the fluoridation effect of enamel against demineralization. |
| preparation | at present, the main methods for preparing calcium glycerophosphate include phosphoric acid method, phosphorus salt method, phosphorus oxychloride method, oil residue method, etc. These methods are complicated, and the purification process of the target product calcium glycerophosphate is complicated and difficult to control. Using the method of calcium hydroxide and glycerophosphate neutralization, the purification process of calcium glycerophosphate was avoided by ethanol extraction. The reaction equation is as follows: The process flow is as follows: |
| content analysis | accurately weigh about 2g of the sample after drying at 150 ℃ for 4h, and dissolve in 100ml of water and 5ml of dilute hydrochloric acid test solution (TS-117). Transfer this solution into a 250ml volumetric flask, and shake well after constant volume with water. Absorb 50.0ml of the solution into a suitable container and add 50ml of water. Under stirring (preferably with a magnetic stirrer), about 30ml of 0.05mol/L EDTA disodium is added dropwise from the burette, 15ml of sodium hydroxide test solution (TS-224) and 300mg of hydroxynaphthol blue indicator are added further, and then the dropwise addition is continued to the blue end point. Disodium EDTA per ml0.05mol/L is equivalent to 10.51mg of calcium glycerophosphate (C3H7CaO6P). |
| toxicity | ADI 0~70 mg/kg (total phosphate in terms of phosphorus, FAO/WHO,2001);GRAS(FDA, 181.29, 182.5201, 182.8201, 184.1201,2000). |
| usage limit | GB 14880-94: beverage 1.6~3.2; Cereals and their products 1.6~3.2; Infant food 3~6(mg/kg, all calculated as calcium). |
| use | nutritional medicine for preparing compound preparations. Nutritional supplements; calcium fortifier for food; stabilizer. Used for baby food, bread, baking powder, wheat flour and noodles. The addition amount is less than 1% (in terms of Ca). |
| production method | add metaphosphoric acid or sodium dihydrogen phosphate to glycerin, heat to obtain glycerophosphoric acid, and then add lime milk to neutralize. It is obtained by neutralizing glycerophosphate with calcium hydroxide or calcium carbonate. |
Supplier List
CAS: 27214-00-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 27214-00-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 27214-00-2
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 27214-00-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 27214-00-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 27214-00-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 27214-00-2
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 27214-00-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025



